Nonvascular benefits of aspirin – its role in cancer
Aspirin for cancer prevention in hereditary colorectal cancer (Lynch syndrome) – the CAPP2 study
CAPP2 is an international, double-blind, randomised, placebo-controlled, registry-based study of cancer prevention in people with Lynch syndrome. Lynch syndrome is a hereditary genetic condition which increases a person’s risk of developing certain cancers (bowel, endometrial, ovary, stomach, pancreas, ureter and renal). The 10 year follow up of CAPP2 gives a long- term assessment of the effect of daily aspirin for cancer prevention in this higher risk population. The study follows 861 patients (mean age 45 years) randomly allocated to receive aspirin 600 mg daily or placebo and monitors cancer outcomes for 10-20 years. The primary outcome measure was colorectal cancer.
In the group receiving aspirin 40 out of the 427 people (9%) developed colorectal cancer in comparison with the 58 (13%) of the 434 taking placebo. An intention-to- treat Cox proportional hazards analysis demonstrated the protective effect of aspirin against colorectal cancer (HR 0.65 [95% CI 0.43-0.97]; p=0.035). The number needed to treat was 24.
Non-colorectal Lynch syndrome cancers were found to be the same in each group. It is interesting that in this relatively young cohort no difference in significant adverse events was observed between the two groups and no significant difference in compliance was noted.
The colorectal cancer protective effect of aspirin in the CAPP2 study took about 5 years to be seen and lasted throughout the observed period, up to 20 years for some participants.
This adds to evidence from over 100 observation studies, carried out since Kune and colleagues first reported a protective effect of aspirin in colorectal cancer, demonstrating aspirin’s cancer prevention benefits.
The authors state:
“There is now a strong case for prescribing aspirin to young adult carriers of a germline DNA mismatch repair gene defect.”
The CaPP3 study is currently ongoing having closed to recruitment in March 2019 and it is hoped that it will inform the best dose of aspirin to use to achieve cancer prevention whilst minimising adverse events
For further information please see:
Burn J, Sheth H, Elliott F et al. Cancer prevention with aspirin in hereditary colorectal cancer (Lynch syndrome), 10-year follow-up and registry-based 20-year data in the CAPP2 study: a double-blind, randomised, placebo-controlled trial. Lancet 2020; 395:1855-63.
Aspirin for Lynch syndrome: a legacy of prevention
In this comment article on Burns et al 2020 [above] the authors explain that some Lynch syndrome-associated colorectal cancers skip the precursor adenoma phase and would therefore not be managed by colonoscopy making a chemoprevention approach even more important.
The Authors point out that in this preplanned update to CAPP2 all participants have passed the 10 year follow up stage and that the data gives us some useful insights:
- A robust reduction in colorectal cancer incidence in Lynch syndrome carriers taking aspirin at a dose of 600 mg/day is seen.
- The benefit emerged after 5 years of taking aspirin – this delayed effect is consistent with findings from studies in non-Lynch syndrome populations.
- A ‘legacy’ effect was seen in Lynch syndrome carriers who only took aspirin for a finite period [on average just over 2 years] in which a reduction in colorectal cancer incidence was still observed 20 years later.
- Fewer endometrial cancers were reported in the female Lynch syndrome carriers but this did not reach statistical significance.
They discuss the ‘legacy’ effect in the context of clinical trials such as ASPREE which show the difficulties and lack of benefit of using aspirin in elderly populations.
The authors question if there is a window of opportunity, from an age perspective, when aspirin can be used with a lower risk of side effects but a lasting impact on cancer incidence sustained long after individuals stop taking aspirin.
The authors state:
“We also eagerly await data from the CaPP3 trial of 1882 Lynch syndrome carriers, which will address the primary question about whether lower aspirin doses (100mg/day and 300mg/day)
are non-inferior to 600mg/day in reducing Lynch syndrome-associated colorectal cancer incidence. Until then, CAPP2 provides a compelling rationale for broader aspirin use in most Lynch syndrome carriers with dosing and duration tailored according to individual risks and benefits.”
For further information please see:
Yurgelun MB and Chan AT. Aspirin for Lynch Syndrome: a legacy of prevention. Lancet 2020; 395: 1817-1818.
Aspirin and primary prevention of colorectal and other digestive tract cancer updated 2019 meta-analysis
This meta- analysis is the result of a systematic review of all the observational studies published until March 2019 investigating the potential reduced risk of colorectal and other digestive tract cancers in people taking aspirin. The researchers found that regular aspirin use resulted in a reduces risk of colorectal cancer, squamous-cell oesophageal cancer, adenocarcinoma of the oesophagus and gastric cardia, stomach cancer, hepato-biliary tract cancer and pancreatic cancer but not head and neck cancer. Interestingly case-control studies showed a stronger association between aspirin use and reduced cancer risk than cohort and nested case-control studies.
In the case of colorectal cancer taking an aspirin dose of 75-100 mg/day gave a 10% risk reduction whilst a dose of 325 mg/day resulted in a 35% risk reduction. High dose aspirin 500mg/day based on a limited number of studies appears to convey a 50% cancer risk reduction.
This favourable effect of aspirin in reducing cancer risk is increased with the duration of use. The cancer prevention effect of aspirin is thought to be due to its inhibition of cyclooxygenase (COX) an enzyme involved in the production of prostaglandins. COX-2 is abnormally expressed in many cancers and is involved in carcinogenesis, tumour growth, apoptosis [programmed cell death] and angiogenesis [formation of blood vessels].
For further information please see:
Bosetti C, Santucci C, Gallus S et al. Aspirin and the risk of colorectal and other digestive tract cancers: an updated meta-analysis through 2019. Annals of Oncology 2020;31(5) 558-568.
Low dose aspirin and hepatocellular carcinoma
This Swedish registry study looked specifically at the long-term effects of low-dose aspirin (160mg or less daily) and the incidence of hepatocellular carcinoma (HCC), liver related death and gastrointestinal (GI) bleeding in people who have chronic hepatitis B or C infection.
They found over a median of 7.9 years of follow-up the incidence of HCC was 8.3% in people not using aspirin but only 4% in those taking low-dose aspirin. As with other studies investigating aspirin’s cancer preventative effects this inverse association was duration dependent. The ten -year liver related mortality was just under 18% on non-aspirin users but only 11% in those taking regular low-dose aspirin. The ten -year risk of gastrointestinal bleeding was not significantly different between the two groups.
For further information please see:
Simon TG, Duberg AS and Aleman S et al. Association of aspirin with hepatocellular carcinoma and liver-related mortality. N Engl J Med 2020; 382(11): 1018-1028.
Inverse relationship found between aspirin use and the risk of developing hepatocellular cancer
This systematic review and dose-response analysis of eight cohort studies with over 2.5 million patients identified a significant inverse relationship between aspirin use and the risk of developing hepatocellular cancer (HCC).
The study found that aspirin doses higher than 100 mg per day did not offer any additional chemoprotective effects and that a dose of 81 mg per day maybe enough to achieve the beneficial effects for most people.
The authors conclude:
That large scale randomised controlled trials will be required to provide proof of aspirins use in the chemoprevention of HCC and that care needs to be taken over the potential for bleeding side effects especially in people with chronic liver disease where bleeding from oesophageal varices or portal hypertensive disease are common clinical complications.
For further information please see:
Wang S, Yu Y, Ryan PM et al. association of aspirin therapy with risk of hepatocellular carcinoma: a systematic review and dose-response analysis of cohort studies with 2.5 million participants. Pharmacological Research Jan 2020; 151, https://doi.org/10.1016/j.phrs.2019.104585
Aspirin to prevent gastrointestinal cancer- but recent trial data do not fit
In a letter to the editor Jacobsen et al highlight the quality and favourable results of the metanalysis conducted by Bosetti et al 2020 [above]. They also report on an observational study by Simon et al 2020 [also above] which found a decreased incidence in hepatocellular carcinoma in people taking aspirin.
Jacobsen and colleagues compare the findings from Bosetti et al’s meta-analysis with three large randomised controlled trials (RCTs) looking at the primary prevention of cardiovascular disease with low-dose aspirin that were published in 2018 and included cancer-related outcomes. The ASCEND trial (with a diabetic population) found no difference in the incidence of gastrointestinal cancer between low-
dose aspirin and placebo groups after a mean follow up of 7.4 years. ASPREE with its elderly population found increased cancer mortality in the aspirin arm of the study after a median of 4.7 years. ARRIVE, with a population of people at moderate risk of cardiovascular disease found the incidence of colorectal cancer was lower in the placebo group when compared with the low-dose aspirin group at a median 5 year follow up (the number of events in this study were however very low).
The writers of the letter question if the ‘antiplatelet’ dose of aspirin used in the cardiovascular primary prevention RCTs is enough to allow for the anti- inflammatory properties of aspirin that maybe required for cancer prevention. They also question if selection bias may have a role to play in the observational studies, for example those taking aspirin for CVD prevention may also minimise smoking and alcohol and participate in colon cancer screening programmes.
They conclude:
“The entirety of evidence may continue to support aspirin’s chemoprevention effects. However, the results of these three high-quality contemporary trials, with their combined 47140 participants, do not fit this narrative, and should give us pause.”
For further information please see:
Jacobsen AP, Khiew YC, Blumenthal RS and Martin SS. Aspirin to prevent gastrointestinal cancer – but recent trial data do not fit. Annals of Oncology https://doi.org/10.1016/j.annonc.2020.05.002
Aspirin in cancer treatment – the Add-Aspirin trial
Support for the concept of aspirin as an add on therapy for cancer treatment comes from preclinical data, epidemiological studies and meta-analysis of randomised controlled cardiovascular trials. The Add- Aspirin trial has been set up to explore the role of aspirin in the treatment of gastro-oesophageal, colorectal, prostate and breast cancer. Data so far indicates that aspirin is well tolerated after cancer treatment and with its low cost it is hoped it will have a global impact on improving cancer outcomes.
The researchers explain:
“The Add-Aspirin study is designed to assess the effect of aspirin on the prevention of metastases and disease recurrence following radical cancer therapy in high risk individuals.”
For further information please see:
Add-Aspirin Trial Management Group. Joharatnam-Hogan N, Cafferty F, Hubner R et al. aspirin as an adjuvant treatment for cancer: feasibility results from Add-Aspirin randomised trial. The Lancet Gastroenterology and Hepatology.2019; 4(11): 854-862
A meta-analysis exploring the effect of aspirin use on gastric cancer incidence and survival
This systematic review and meta-analysis, which included nearly two million patients across 33 studies sought to estimate risk reduction with aspirin use on gastric cancer incidence and mortality.
Helicobacter pylori infection causes inflammation within the stomach and is a common cause of cancer worldwide. Aspirin use is protective because of its anti- inflammatory and antiplatelet functions.
Aspirin use was found to be associated with a reduced gastric cancer incidence and mortality and this effect was greater in Asian and North American populations, noncardiac cancer and where duration of use was less than 5 years.
For further information please see:
Niikura R, Hirata Y, Hayakawa Y et al. Effect of aspirin use on gastric cancer incidence and survival: A systematic review and meta-analysis. JGH Open 4;2; https://doi.org/10.1002/jgh3.12226.
Low-dose aspirin reduces the risk of gastric and oesophageal cancer
This study used a UK population-based primary care database to quantify the association between low-dose aspirin (75-300mg/day) use and the risk of developing a gastric or oesophageal cancer. A cohort of 223640 new low-dose aspirin users were identified between January 2005 and December 2015 and these were matched with a cohort of non-aspirin uses.
This study is set within the general population and adds to existing evidence that daily low-dose aspirin use appears to reduce the risk of developing gastric or oesophageal cancer. This effect is thought to be due to the inhibitory effect aspirin has on both the development of existing tumours and metastasis. The authors call for further population-based research to help clarify aspirins effect.
The authors state that:
“Our results indicate that use of low-dose aspirin is associated with a 54% reduced risk of gastric cancer and a 41% reduced risk of oesophageal cancer as supported by mechanistic data”
For further information please see:
Luis A, Rodríguez G, Soriano-Gabarró M, Vora P et al. Low-dose aspirin and risk of gastric and oesophageal cancer: a population- based study in the United Kingdom using The Health Improvement Network. International Journal of Cancer. April 2020 https://doi.org/10.1002/ijc.33022.
Extracts from the 2019 International Aspirin Foundation Scientific Conference, Rome, 2019: benefits and risks of antithrombotic therapy for cardiovascular disease prevention
Benefits and risks of antithrombotic therapy in primary prevention was one of the many topics discussed during our Scientific Conference, where international experts gathered to discuss and debate ‘Incorporating other benefits of low-dose aspirin in the benefit/risk equation.’
Below are the speaker biographies and their extract from the conference report.
- Paola Patrignani (University of Chieti)
Mechanisms underlying the non-vascular effects of low-dose aspirin - Ruth Langley (University College London)
Clinical trial evidence supporting a cancer chemopreventive effect of low-dose aspirin
Professor Paola Patrignani
Professor of Pharmacology at the Catholic University School of Medicine in Rome, Italy.

Professor of Pharmacology, Department of Neuroscience, Imaging and Clinical Sciences, Section of Cardiovascular and Pharmacological Sciences, “G. d’Annunzio” University, Chieti, Italy.
Paola Patrignani was graduated at the Faculty of Biological Sciences, “La Sapienza” University of Rome (Italy). Then, she followed a doctoral training in Pharmacology, at Catholic University of Rome, Italy. She worked for 2 years as Postdoctoral Research Fellow at Le Centre Hospitalier of Laval University (Quebec, Canada) and at the Department of Pharmacology of Merck Frosst (Kirkland, Quebec, Canada).
She continued her academic career at “G. d’ Annunzio” University, School of Medicine, Chieti, Italy, where she is currently Professor of Pharmacology and Head of the Laboratory of Systems Pharmacology and Translational Therapeutics at the Center of Excellence on Aging and Translational Medicine (CeSI-MeT).
In 2009 she was Guest professor of Pharmacology at Goethe-Universität Frankfurt am Main (Germany). She is a member of the Italian Society of Pharmacology and she received the Upjohn Award (1981), the “Henry Christian” Award (1991) and was the winner of the 2018 International Aspirin Foundation Senior Science Award. In 2013, she received a special prize awarded by the Committee for the Promotion of Female Entrepreneurship of Abruzzo Region (Italy).
She is one of the Top Italian Women Scientists (TWIS) which includes the scientists with high impact publications. Her scientific activity is documented by 170 peer-reviewed publications in international journals ranked in the Journal Citation Reports, and 21 chapters in national-international books. Her cumulative citation index (H-index) is 56, 11831 citations.
Mechanisms underlying the non-vascular effects of low-dose aspirin
Prof. Paola Patrignani (‘G. d’Annunzio’ University of Chieti, Italy) explained the mechanisms underlying the non- vascular effects of low-dose aspirin. The data for a chemopreventive role for low-dose aspirin against colorectal cancer (CRC) and other cancers is growing. This effect appears to be from the prevention of early neoplastic transformation within the gut and an anti-metastatic action. The antiplatelet activity of low-dose aspirin can mostly explain both of these effects. Aspirin acts by acetylating platelet COX-1 at a critical serine residue (Ser-529) close to the catalytic site of this enzyme.
As a result of in vitro studies, using supratherapeutic concentrations, various mechanisms for aspirin anti-cancer action have been proposed:
(a) Inhibition of nuclear factor kappa B (NF-kB) signalling
(b) Interruption of extracellular signal-regulated kinases (ERK)
(c) Inhibition of Wnt/b-catenin signalling
(d) Capacity of aspirin to acetylate extra-COX proteins, e.g. p53
(e) Activation of adenosine monophosphate-activated protein kinase.
Low-dose aspirin can also partially acetylate rectal mucosal COX-1, associated with the reduction in PG E2 and phosphorylated S6 (p-S6) levels. This effect might interfere with early colorectal carcinogenesis [51, 52].
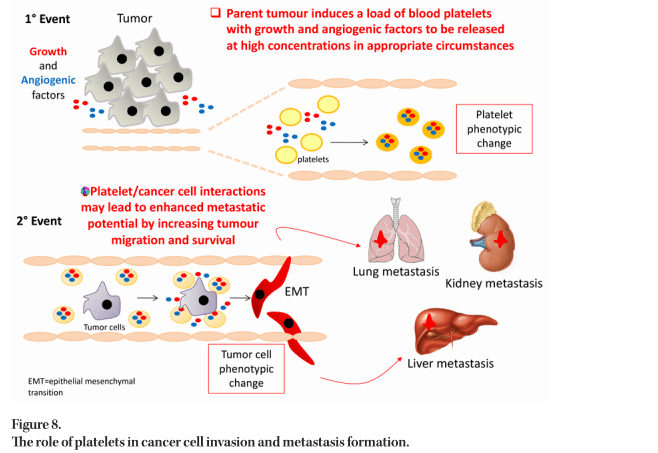
Platelets sustain cancer cell invasion and metastasis formation by supporting the development of epithelial– mesenchymal transition (EMT), cancer cell survival in the bloodstream and the enhancement of tumour cell adhesion to the endothelium facilitating arrest and extravasation. Platelets also contribute to tumour escape from immune elimination.
Aspirin, by inhibiting platelet activation triggered by GI mucosal lesions, restrains the development of chronic inflammation, which is crucial in cancer development. Aspirin can also acetylate COX-1 expressed in colorectal mucosa leading to changes in the mucosal phenotype.
The interaction of cancer cells with platelets results in the following molecular and functional consequences:
(a) Induction of COX-2 in CRC cells (a hallmark of cancer)
(b) Induction of EMT in CRC cells promoting migration and metastatic potential (c) Induction of a prothrombotic phenotype in cancer cells
(d) Enhanced systemic biosynthesis of TXA2 [53, 54].
Evidence is now accumulating to support the hypothesis that activated platelets contribute to CRC development and metastatic spread by direct cell to cell interactions with stromal cells and cancer cells, together with the release of different lipid and protein mediators and microvesicles (Figure 8). Platelets can also uptake proteins and genetic material that is present within the bloodstream. Evaluating the proteomics and transcriptomics signature of platelets and platelet-derived microparticles may present a new strategy for developing biomarkers for early cancer detection and for therapeutic drug monitoring in cancer chemotherapy.
Professor Ruth Langley
 Professor Ruth Langley is a Medical Oncologist specializing in the design and management of oncology clinical trials based at the MRC Clinical Trials Unit at University College London where she leads the Cancer Group. Professor Ruth Langley has worked in a number of tumour areas including colorectal, lung and gastro-oesophageal cancer coordinating a series of trials and associated translational studies. She has a particular interest in re-purposing established medicines as cancer therapeutics, as well as cancer prevention, and is Chair of the UK Therapeutic Cancer Prevention Network. She has led the investigation of the use of transdermal oestrogen as a treatment for prostate cancer (PATCH studies). A major focus of her recent work has been the development of an international trial to assess the effect of aspirin as an adjuvant agent in several common solid tumours (the Add-Aspirin Trial) which is recruiting patients from the UK, Ireland and from several sites across India. The Add-Aspirin trial is part of a Cancer Research UK funded initiative the AsCaP collaboration to investigate the mechanisms underlying the anti-cancer effects of aspirin. She is a faculty member of the Indian CReDO (Collaboration for Research methods Development in Oncology) programme.
Professor Ruth Langley is a Medical Oncologist specializing in the design and management of oncology clinical trials based at the MRC Clinical Trials Unit at University College London where she leads the Cancer Group. Professor Ruth Langley has worked in a number of tumour areas including colorectal, lung and gastro-oesophageal cancer coordinating a series of trials and associated translational studies. She has a particular interest in re-purposing established medicines as cancer therapeutics, as well as cancer prevention, and is Chair of the UK Therapeutic Cancer Prevention Network. She has led the investigation of the use of transdermal oestrogen as a treatment for prostate cancer (PATCH studies). A major focus of her recent work has been the development of an international trial to assess the effect of aspirin as an adjuvant agent in several common solid tumours (the Add-Aspirin Trial) which is recruiting patients from the UK, Ireland and from several sites across India. The Add-Aspirin trial is part of a Cancer Research UK funded initiative the AsCaP collaboration to investigate the mechanisms underlying the anti-cancer effects of aspirin. She is a faculty member of the Indian CReDO (Collaboration for Research methods Development in Oncology) programme.
Clinical trial evidence supporting a cancer chemopreventive effect of low-dose aspirin
Prof. Ruth Langley (University College London, UK) presented an overview of the clinical trial evidence for aspirin’s nonvascular, cancer chemopreventive effect.
The talk was divided into two areas:
(a) Aspirin for the prevention of primary cancers and progression of pre-malignant lesions
(b) Aspirin for the prevention of metastases and recurrence after radical therapy.
The evidence for aspirin’s nonvascular effects comes from pre-clinical work, epidemiological studies and randomised trials including those that were initially designed to assess the vascular effects of aspirin [55–57].
In terms of primary prevention, recent studies include SeaFOod where mean colorectal adenomas per patient were reduced by aspirin particularly right-sided, serrated lesions supporting previous data [58] and the AspECT trial which showed that high dose PPIs and aspirin compared to low dose PPI/no aspirin for patients with Barrett’s oesophagus increased time to event defined as high-grade dysplasia, oesophageal adenocarcinoma and all-cause mortality [59]. The CAPP2 trial [60] has already demonstrated that patients with Lynch syndrome benefit from aspirin and results are awaited from the CAPP3 trial which recently completed recruitment and is focussed on the dose of aspirin required for anti-cancer effects.
Results from the ASPREE trial [61] (aspirin in an elderly population) were unexpected. The primary outcome measure was a composite of death, dementia and permanent physical disability, with a follow up of 4.5 years 21.5 versus 21.2 events per 1,000 person-years were seen with aspirin compared to placebo (HR 1.01, 95% CI 0.92–1.11). However, an increase in mortality with aspirin was noted which has been attributed to an increase in the risk of metastatic cancer but not incident cancers overall. This might be accounted for by aspirin unmasking malignant disease through an increased risk of bleeding particularly as the incidence and severity of bleeding with aspirin increase with age.
A number of clinical trials are also underway to evaluate the role of aspirin as an adjunct therapy for colorectal, breast and other cancers (ASCOLT, ASPIRIN, US Aspirin Breast Cancer (ABC) trial, PIK3CA based trials and the Add-Aspirin trial). The PIK3CA mutation trials are based on epidemiological data that aspirin after a diagnosis of CRC is most effective if the cancer has a PIK3CA mutation [62]. The Add-Aspirin trial is recruiting patients from the UK and India and aims to assess the addition of either 100 or 300 mg of aspirin after standard primary therapy for early-stage breast, colorectal, gastro-oesophageal and prostate tumours. Over 7,000 patients have been recruited to this trial and will be followed for at least 5 years with long-term passive follow-up via the National Cancer Intelligence Network (NCIN) in the UK [63]. Adherence in this study has been good with 95% of participants taking six to seven of the tablets per week. Data from the run-in period of the trial (aspirin 100 mg open-label for 8 weeks) from over 2,000 patients has shown a low toxicity rate with only 13/2,253 (0.6%) of the participants having had a grade 3 toxicity.
Prof. Langley concluded by stressing the importance of long-term follow-up in both primary and adjuvant aspirin cancer trials and noted the importance of electronic health records for this work. She also commented that the benefit/risk ratio for aspirin in terms of cancer prevention is likely to favour aspirin use in a ‘younger’ (middle-aged rather than elderly) population. Cancer Research UK (CRUK) recently funded an international collaboration to further explore the role of aspirin in cancer prevention with the aim of understanding the mechanism of action. Finally, the concept of cancer chemoprevention in primary, secondary and tertiary approaches needs wider promotion and greater public awareness.












