Summary of European guidelines for aspirin
August 2023
The purpose of this document is to keep a working list of European guidelines with information on aspirin. The guidelines themselves should be read for further information.
European guidelines make the following recommendations concerning aspirin and VTE prophylaxis:
- We recommend the use of aspirin as an option for venous thromboembolism (VTE) prevention after total hip arthroplasty, total knee arthroplasty and hip fracture surgery (Grade 1B).
- We suggest the use of aspirin for VTE prevention after total hip arthroplasty, total knee arthroplasty and hip fracture surgery (high-risk procedures) in patients without high VTE risk (Grade 2C).
- We suggest the use of aspirin for VTE prevention after low-risk orthopaedic procedures in patients with a high VTE risk or other high-risk orthopaedic procedures in patients without a high VTE risk (Grade 2C).
- We suggest the use of aspirin for VTE prevention after total hip arthroplasty, total knee arthroplasty and hip fracture surgery in patients with an increased bleeding risk (Grade 2C).
- We suggest the use of aspirin for VTE prevention after total hip arthroplasty or total knee arthroplasty in a rapid recovery (fast-track) programme (Grade 2C).
- We recommend combining aspirin with intermittent pneumatic compression (IPC) devices for VTE prevention after total hip arthroplasty, total knee arthroplasty and hip fracture surgery (Grade 1C).
- We recommend no pharmacological VTE prevention after low-risk orthopaedic procedures in patients without high VTE risk (e.g. knee arthroscopy) (Grade 1C).
- No recommendation can be made concerning dose and duration of aspirin treatment and patient selection.
- We do not recommend aspirin for thromboprophylaxis in general surgery (grade 1C). However, this type of prophylaxis could be interesting especially in low income countries (Grade 2C) and adequate large-scale trials with proper study designs should be carried out (Grade 1C)
David R. Anderson, Gian Paolo Morgano, Carole Bennett, Francesco Dentali, Charles W. Francis, David A. Garcia, Susan R. Kahn, Maryam Rahman, Anita Rajasekhar, Frederick B. Rogers, Maureen A. Smythe, Kari A. O. Tikkinen, Adolph J. Yates, Tejan Baldeh, Sara Balduzzi, Jan L. Brożek, Itziar Etxeandia- Ikobaltzeta, Herman Johal, Ignacio Neumann, Wojtek Wiercioch, Juan José Yepes-Nuñez, Holger J. Schünemann, Philipp Dahm; American Society of Hematology 2019 guidelines for management of venous thromboembolism: prevention of venous thromboembolism in surgical hospitalized patients. Blood Adv 2019; 3 (23): 3898–3944. doi: https://doi.org/10.1182/bloodadvances.2019000975
Primary prevention of cardiovascular disease
Key Message: “Antiplatelet therapy is not recommended in individuals free from CVD, due to the increased risk of major bleeding.” European Society of Cardiology (ESC) 2016
The 2016 European guidelines on CVD prevention review the evidence for antiplatelet therapy in individuals without CVD and conclude that current evidence does not support the use of aspirin in those without CVD due to the risk of a major bleed.
For more information see:
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/CVD-Prevention-in-clinical-practice-European-Guidelines-on
The ESC 2019 guidelines on diabetes (DM), pre-diabetes, and cardiovascular disease state:
- “Patients with DM and symptomatic CVD should be treated no differently to patients without DM
- In patients with DM at moderate CV risk, aspirin for primary prevention is not recommended
- In patients with DM at high/very high risk, aspirin may be considered in primary prevention
For more information see:
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Diabetes-Pre-Diabetes-and-Cardiovascular-Diseases-developed-with-the-EASD
In addition, the ESC 2019 diabetes guidelines identify the following gaps in the evidence relating to platelet use in people with diabetes:
- Type 1 diabetes and CVD prevention [in vivo platelet activation has been reported]
- Body mass and antiplatelet responsiveness especially in those with diabetes and obesity where higher dose strategies need investigation
- Are antithrombotic effects similar in pre-DM and DM?
The ESC does not recommend aspirin for healthy people over 70 following results of the ASPREE trial.
https://www.escardio.org/The-ESC/Press-Office/Press-releases/aspirin-should-not-be-recommended-for-healthy-people-over-70
The ESC state in a press release following results of ARRIVE that the benefits of aspirin in primary prevention remain unclear.
https://www.escardio.org/Congresses-&-Events/ESC-Congress/Congress-resources/Congress-news/benefit-of-aspirin-for-primary-prevention-of-cardiovascular-events-remains-unclear-arrive-trial-results
In another press release following the ASCEND trial the ESC state that the bleeds and benefits are balanced in people with diabetes using aspirin for primary CVD prevention and that no cancer benefit was found in this study.
https://www.escardio.org/The-ESC/Press-Office/Press-releases/bleeds-and-benefit-with-aspirin-balanced-in-patients-with-diabetes-and-no-effect-on-cancer
Secondary prevention of cardiovascular disease
The 2020 ESC guidelines for the management of acute coronary syndromes, in patients presenting without persistent ST-segment elevation, recommends that people [without atrial fibrillation] undergoing percutaneous coronary intervention are given aspirin, unless contraindicated, at a loading dose of 150-300 mg once daily and a maintenance dose of 75-100 mg once daily. They recommend a P2Y12 receptor inhibitor is given in addition to low-dose aspirin for the first 12 months
For further information see:
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Acute-Coronary-Syndromes-ACS-in-patients-presenting-without-persistent-ST-segm
The 2019 ESC guidelines for the diagnosis and management of chronic coronary syndromes recommended aspirin for CVD event prevention at a dose of 75-100 mg daily in patients with previous MI or revascularization. In addition, aspirin 75-100 mg daily can also be considered for people without a history of MI or revascularization but where there is evidence of coronary artery disease using imaging tests.
For further information see:
https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Chronic-Coronary-Syndromes
The 2016 European Guidelines for CVD prevention in clinical practice make the following recommendations for antiplatelet therapy (see table below). This is a based on a careful appraisal of risks versus benefits of aspirin or other antiplatelet therapy. The guidelines state that there is a gap in the evidence concerning new antiplatelet drugs in patients with stable coronary artery disease as well as a gap in the understanding about the use of new antiplatelet drugs used in combination with anticoagulation treatment.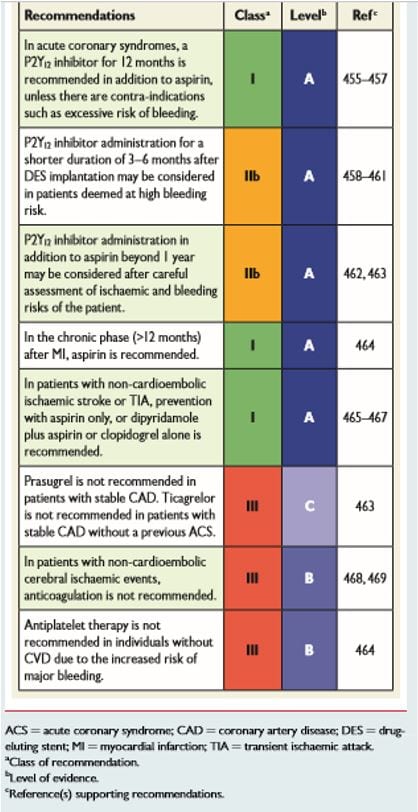
Source 2016 European guidelines for cardiovascular disease prevention
For further information please see the full guidance at:
Primary prevention of cancer
There is a European Code Against Cancer code which covers healthy lifestyle, work place safety, vaccination programmes and cancer screening.
https://cancer-code-europe.iarc.fr/index.php/en/
Aspirin and pre-eclampsia
The International Federation of Gynecology and Obstetrics recommend that women identified as high risk of pre-eclampsia during first trimester screening should be given aspirin prophylaxis (150mg at night from 11-14 weeks gestation until delivery or the diagnosis of pre-eclampsia). They do not advocate a policy of low-dose aspirin for all pregnant women.
Poon LC, Shennan A, Hyett JA et al The International federation of gynecology and Obstetrics (FIGO) initiative on pre-eclampsia: a pragmatic guide for first-trimester screening and prevention. Int J Gynecol Obstet 2019; 145 (Suppl.1) 1-33. @
https://obgyn.onlinelibrary.wiley.com/doi/epdf/10.1002/ijgo.12802











